Environmental Cleanup & Restoration: CASE STUDY
Foundry Cove in the Hudson River, New York
Adapted from: Foundry Cove: History of a Polluted Site and Its Restoration by Jeffrey Levinton
and Josepha Kurdziel, SUNY Stony Brook:
http://life.bio.sunysb.edu/marinebio/foundryframe.html
History of Metal Pollution in Foundry Cove
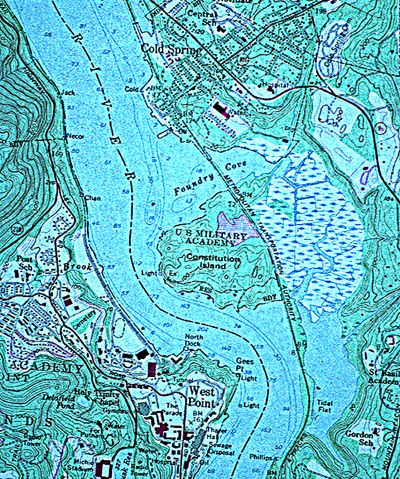 Foundry Cove is situated in the Village of Cold Spring, in Putnam County, NY, approximately 54
miles north of Battery Park, NYC (Fig 1). Foundry Cove is a well defined inlet of the Hudson
River. This cove contains freshwater marshes and mud flats and is tidally influenced (Fig. 2). A
railroad trestle divides the cove into east and west; flow from the Hudson River into West
Foundry Cove is unrestricted, while flow into East Foundry Cove is restricted to a 65 foot wide
passage under the railroad
trestle. The water is
generally fresh but the
salinity may reach 2-6 parts
per thousand in periods of
low fresh water flow.
Foundry Cove is situated in the Village of Cold Spring, in Putnam County, NY, approximately 54
miles north of Battery Park, NYC (Fig 1). Foundry Cove is a well defined inlet of the Hudson
River. This cove contains freshwater marshes and mud flats and is tidally influenced (Fig. 2). A
railroad trestle divides the cove into east and west; flow from the Hudson River into West
Foundry Cove is unrestricted, while flow into East Foundry Cove is restricted to a 65 foot wide
passage under the railroad
trestle. The water is
generally fresh but the
salinity may reach 2-6 parts
per thousand in periods of
low fresh water flow.
The Marathon
Battery Company facility in
Cold Spring, NY, was
located near Foundry Cove.
The plant was constructed in
1952 by the U.S. Army
Corps. From 1952 through
1979 this facility
manufactured nickel-cadmium (Ni-Cd) batteries,
initially for military
contracts. The plant was
later owned by several
private companies
(Sonotone Corporation,
Clevite Corporation, and
later Gould Incorporated)
that produced batteries for
commercial use.
Fig. 1. Area map showing location of Foundry Cove on the Hudson River.
The battery manufacturing process requires the use of concentrated metal nitrate solutions
that result in dilute waste solutions and metal precipitates. Both nickel and cadmium were used in
large quantities; for a brief time, cobalt was used as an additive. The plant effluent was a fine
suspension of nickel and cadmium hydroxides, in a pH range of 12 - 14, at a volumetric flow rate
averaging 50 - 100 gallons/minute. The effluent usually contained from 10 - 100's ml/l suspended
Ni and Cd hydroxides, depending upon production values. The total waste water output ranged
from 100,000 - 200,000 gallons/day (Klerks 1987).
 Fig. 2. Foundry Cove,
foreground, with Constitution
Marsh Audubon Sanctuary at
Center Left. South Cove is at the
Rear
Fig. 2. Foundry Cove,
foreground, with Constitution
Marsh Audubon Sanctuary at
Center Left. South Cove is at the
Rear
Waste water from the
manufacturing process was
initially discharged into the
Hudson River through the
Cold Spring sewer system, but
approximately 10% was
discharged into a bypass
system emptying directly into
East Foundry Cove. In 1965,
the NY State Department of
Health concluded that the village of Cold Spring's sewage treatment system could not handle the
plant's industrial waste water; the battery company was ordered to disconnect from the sewer
system after which all waste was discharged directly into East Foundry Cove. After maximum
discharge limits were set in 1971, waste waters were again discharged via the sewer system into
the Hudson River. During manufacturing operations, a total of 179,105 kg of cadmium hydroxide
was discharged. Of this amount, 51,004 kg of particulate Cd, and 1,569 kg of soluble cadmium
were discharged directly into East Foundry cove; the remainder was discharged in the Hudson
River (Klerks 1987). This earned
Foundry Cove the dubious distinction of
being "the most cadmium polluted site in
the world".
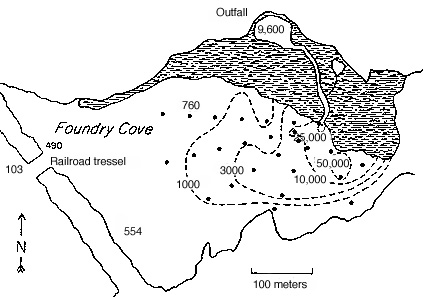
Fig. 3. Distribution of cadmium in surface
sediments in East Foundry Cove in 1974
Fig. 4. Distribution of cadmium in surface
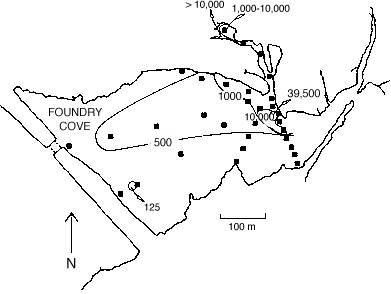 sediments in East Foundry Cove in 1983
sediments in East Foundry Cove in 1983
In 1971, state officials detected
high cadmium levels in East Foundry
Cove in violation of the Clean Water Act
of 1970. A civil law suit filed against
Marathon Battery Company resulted in
the dredging of all sediment exceeding
900 mg/g Cd based on wet weight. In
1972 - 1973, this dredging removed 10%
of Cd released into Foundry Cove. These contaminated sediments (90,000 m3) were buried in a
clay-lined, underground vault on the plant property. Extremely high Cd and Ni concentrations
were found in sediments in subsequent years, despite the dredging; up to 50,000 and 11,000 �g
Cd and Ni per g dry weight sediment (Hazen & Kneip 1979, Occhiogrosso et al. 1979). In 1975,
about 30% of the cove still had surface Cd levels in excess of 1000 ppm. In 1979, the Marathon
company closed the plant and relocated. Merchandise Dynamics purchased the plant in 1980 for
use as a book storage facility. In that same year Congress enacted the comprehensive
Environmental Response, Compensation, and Liability Act (CERCLA) to address the cleanup of
the nation's hazardous waste sites.
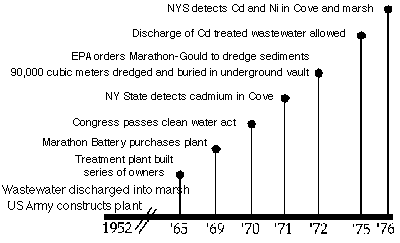
Fig 5. Timeline for the history of cadmium pollution of Foundry Cove
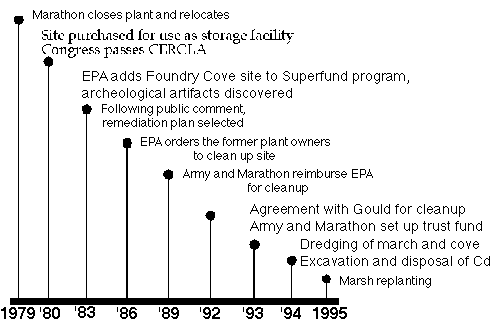
Fig 6. Timeline for the cleanup of Foundry Cove
Investigations into the former battery plant began again in 1983 when the NY State
Department of Environmental Conservation (DEC) sampled soils on the property as well as marsh
sediments in Foundry Cove. At that time, only 8% of the total area of Foundry Cove contained
surface sediment Cd levels in excess of 1000 �g/g dry weight; the values for Ni and Co were
much lower, but spatially strongly correlated with the Cd values. A remediation plan was designed
and managed by the U.S. Environmental Protection Agency (EPA). Due to the severity and extent
of cadmium contamination, the EPA added Foundry Cove to the Superfund program, a national
priorities list of abandoned hazardous waste sites requiring long-term cleanup. In 1986, the EPA
divided the site into three geographical areas and following a period of public comment, a
remediation plan for the
three areas was drafted.
Dredging and excavation
activities began in 1992
and were completed in
1994. Marsh replanting
efforts and the
demolishing of plant
buildings are ongoing.
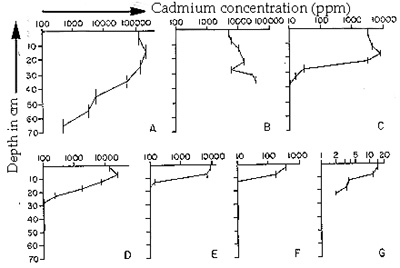
Fig. 7. Cadmium profiles
with depth in the sediment,
as measured in 1983
Unlike some toxic compounds, e.g., PCB's, which can be broken down by natural
processes or through remediation techniques, metals like cadmium cannot be degraded. The
natural reduction of surface Cd concentrations in Foundry Cove from 1971 to 1983 (prior to
EPA's excavation and dredging activities) must be due either to: (1) deposition of new sediment,
(2) transport of metals out of the cove, (3) redistribution of sediments within the cove, or (4)
some combination of these processes. Depth profiles were established in order to determine if
burial had occurred. At the most polluted sites sampled in 1983 (those closest to the battery plant
outfall pipe [sites A - E]), there was a subsurface peak in Cd concentration, suggesting that the
polluted sediment was being covered by new sediment (Knutson et al. 1987). Movement of Cd
out of the cove may also account for a portion of the decreased Cd concentrations. Feeding
processes of invertebrates within the cove will also affect metal distributions in surface sediments
as well as transfer of metals to other parts of the Hudson River ecosystem.
Animal Feeding and Metal Uptake and Bioconcentration
The benthic oligochaete, Limnodrilus hoffmeisteri (Fig. 8), inhabiting cadmium-, nickel-,
and cobalt-polluted Foundry Cove has evolved resistance to these metals (Klerks & Levinton
1989a,b). In survival experiments in which oligochaetes from Foundry Cove and South Cove
(control site) were exposed to sediment with highly elevated metal levels, Foundry Cove worms
survived the 28 day exposure while control worms did not. Second generation offspring of
Foundry Cove worms reared in clean sediment also possess metal resistance.
Fig 8. Cluster of Limnodrilus hoffmeisteri
An increased resistance to a metal can be achieved by a
reduced accumulation of the pollutant. Reduced uptake rates
have been reported for a number of different organisms, e.g.,
bacteria, algae, annelids, and fish (references cited in Klerks &
Bartholomew 1991). But several studies comparing metal
accumulation in populations differing in resistance did not find
reduced uptake rates in resistant populations; some authors even
found increased uptake rates. If resistant individuals have increased metal uptake rates, then they
must possess some physiological mechanism for metal detoxification. The ultimate research goal
is to determine the mechanism(s) by which resistance has evolved in Limnodrilus hoffmeisteri
inhabiting Foundry Cove.
The first objective towards this ultimate goal was to determine whether resistant worms
accumulate less Cd than their sensitive conspecifics from South Cove. Secondly, if metal resistant
and sensitive individuals have similar uptake rates, is resistance achieved by an increased cadmium
detoxification mechanism? The following experiments determined metal uptake rates in resistant
and control populations using the radioisotope 109Cd in water and Foundry Cove sediments.
Limnodrilus hoffmeisteri (Annelida, Oligochaeta, Tubificidae) is a simultaneous
hermaphrodite, which reproduces sexually by cross-fertilization. This oligochaete is a
deposit-feeder and the most common macrobenthic species at both coves (Klerks & Levinton
1989b). Worms and sediment were collected by Ekman grab from both Foundry and South coves.
Worms were then sorted from the >500 �m fraction. Laboratory cultures were set up in
polystyrene dishes with 1 cm layer of sediment and 9 cm of continuously aerated Hudson River
water. Sediments collected from the two coves were sieved to < 500 �m, boiled, washed with
filtered Hudson River water, then frozen and thawed shortly before use. Ground fish food flakes
were added to dishes once per week and cultures were kept at 24C under a 13:11 light:dark
cycle.
It is likely that resistance in Limnodrilus in Foundry Cove evolved mainly in response to
Cd pollution at this site, rather than Ni or Co pollution. This is based on several observations:
these worms accumulate much more Cd than Ni, but they do not accumulate Co at all, and Cd is
generally much more toxic than Ni (Khangarot & Ray 1987). This investigation into the
mechanism(s) underlying the resistance in Limnodrilus in Foundry Cove thus focused on the fate
of Cd in these worms.
Cadmium Uptake from Sediments
Cadmium accumulation in L. hoffmeisteri from Foundry Cove and South Cove was
determined by exposing worms to sediment with different metal levels. These exposure
experiments were set up as a bioassay for comparison of sediment toxicity among populations
(Klerks & Bartholomew 1991). Three replicates of 10 worms each from stock cultures were
exposed to 6 different sediment metal levels (ranging from 15 to 34,000 �g/ g dry sediment) for
28 days. Worms that survived the exposure and laboratory stock worms were collected for metal
analyses (6-10 worms per replicate). These worms were kept in filtered river water for 2 days to
exclude gut contents from analyses. The worms were then rinsed in distilled water, pooled by
replicate, and frozen.
Worms from each replicate were then thawed, transferred to a beaker and dried. Ultrex
grade nitric acid (2 ml) was then added to each beaker, refluxed for 2-4 h at 120C, and
evaporated. This procedure was repeated twice, after which each sample was brought to 5ml
volume with addition of nitric acid. Cadmium concentrations were determined with a Perkin
Elmer 4000 graphite furnace atomic absorption spectrophotometer. Blanks, tissue samples, and
standards were run concurrently; the use of the National Bureau of Standards Oyster Tissue
resulted in values within the range specified for this reference material.
Cadmium Uptake from Solution
To determine Cd accumulation from water, resistant and sensitive worms were exposed in
plastic petri dishes to 8.9 �M (= 1 mg/l) Cd in reconstituted fresh water (pH 7.8-8.0) for 6 days
(Klerks & Bartholomew 1991). The addition of 109Cd resulted in a radioactivity of 22.2 kBq/ml in
the exposure water. Three replicates of 10 worms each from Foundry Cove and South Cove stock
cultures were exposed and survivors collected and frozen at -80C. Thawed samples were later
homogenized in 50 mM Tris-HCl buffer (pH 7.4) using a 50:1 ratio of buffer to tissue. Cadmium
concentrations of each homogenate were determined by gamma counting, using a Beckman 4000
gamma counter with a 3-inch sodium iodide crystal.
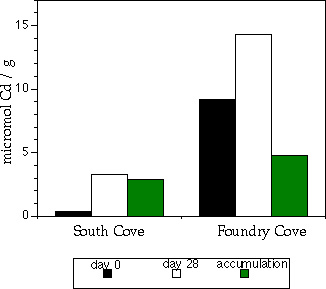
Fig. 9. Uptake of Cd by South Cove worms versus
Foundry Cove worms. Note that Foundry Cove
worms took up and accumulated more Cd.
Results
Cd concentrations of Limnodrilus hoffmeisteri
from the sediment toxicity bioassays did not
differ among groups exposed to sediment with
Cd raging from 5,400 to 34,000 �g-Cd/g-dry
sediment (p > 0.05, ANOVA) for worms from
Foundry as well as South Cove (Fig. 9). These
data were thus pooled to compare Cd
accumulation of Foundry Cove worms with that
of conspecifics from the control area (South Cove). The data provide no evidence for a reduced
Cd accumulation in Foundry Cove worms; worms from Foundry Cove accumulated significantly
more Cd in both sediment and water bioassays.
The data show that Cd-resistance in Limnodrilus from Foundry Cove is not due to a
reduced accumulation of the metal. Worms from Foundry Cove actually accumulate more Cd than
their metal-sensitive conspecifics from South Cove. Since Foundry Cove worms accumulate more
Cd than their sensitive conspecifics, other mechanisms, such as sequestration (binding up the Cd
in a detoxifying compound), must be responsible for the Cd resistance.
Other Animals Are Affected by Cadmium
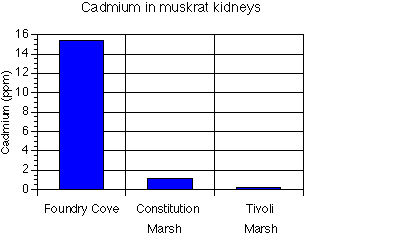
Muskrats were conspicuously rare in Foundry Cove. Cadmium was measured in kidneys and
elevated levels indeed were found compared to muskrats from control areas (Fig. 10a). It was
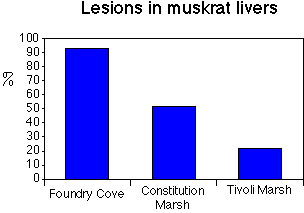
thought that muskrats might have trouble with physiological function so lesions on the liver were
counted in Foundry Cove muskrats, as compared to other marshes (Fig. 10b). Clearly, Foundry
Cove muskrats were having problems, as evidenced by the high incidence of liver lesions.
Fig. 10. (a) Cd in muskrat kidneys, and (b) lesions observed on muskrat livers.

Mitigation and Recovery
In some localities, toxic materials exist at
concentrations high enough to pose potential
health risks. These toxic materials contaminate
water, bind to sediments, and potentially
bioaccumulate in the tissues of plants and
animals. Many toxic materials that are detected
in the environment are not due to new input, but
due to existing compounds recycling between the
water, sediments, atmosphere, and organisms.
Fig. 11. Sign at Marathon Battery Factory Site, 1994
Biological uptake patterns may differ among organisms; on the basis of such differences
toxic materials are grouped into two categories: non-cumulative and cumulative toxic materials.
Non-cumulative toxic materials do not increase in concentration in the body, even if the organism
is chronically exposed to the toxin. Conversely, cumulative toxic materials, tend to increase in
concentration, and are often associated with a specific tissue, e.g., Cd tends to increase over time
in the digestive gland of blue crabs. Such accumulation may lead to food chain magnification, i.e.,
the magnification of toxic materials concentrations across trophic levels when the prey species
possesses a physiological mechanism which concentrates the toxin in a specific tissue and the
predator consumes large quantities of this prey.
Consumption of blue crabs by humans in the Hudson River is restricted because Cd can
impair kidney function even at relatively low concentrations. Higher Cd accumulation in humans
can potentially lead to other serious health risks including bone deformities, cardiovascular and
immune system deficiencies, central nervous system disorders, and lung cancer. Beyond the risks
to human health, high toxic materials contamination also poses ecological concerns by threatening
indigenous wildlife and plant species. Toxic contamination also imposes economic and other
societal effects as remediation is often very costly (beyond a local county or state budget) and the
individuals involved in policy decisions differ in opinion as to the best course of action in
remediation efforts.
Twenty-seven years of Ni-Cd battery production left Foundry Cove contaminated with
179 metric tons of cadmium. In 1983, a remedial investigation of Foundry Cove by the EPA and
the NY State DEC, resulted in EPA declaring Foundry Cove a "superfund site" and millions of
dollars were allocated to the cleanup.
The Marathon Battery site encompasses wetlands, archaeologically and historically
sensitive lands (Indian artifacts and Civil War relics were discovered at the site), a warehouse
facility, and contaminated residential areas (yards). The EPA designed specific treatments to
address the effects of extensive metal contamination.
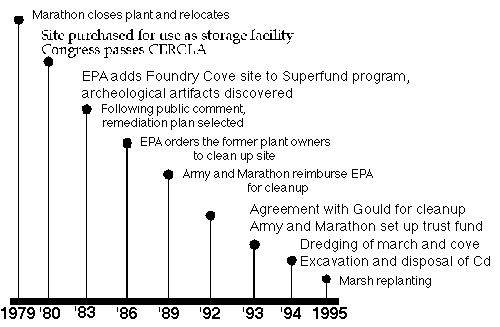
Fig. 12. Timeline of Mitigation
Foundry Cove Timeline 1979-1995
Using Superfund authority, and with the advice of scientists and residents, EPA designed
the following remediation measures: (1) dredging, draining, and treating contaminated sediments
and replanting acres of marshes along Foundry Cove, (2) excavating and treating contaminated
soil in an underground vault on the plant property and tearing down plant buildings and
processing towers, (3) decontaminating and recycling books stored at the plant, and (4)
excavating contaminated soil from residential yards near the site and landscaping these yards. The
EPA settled with the former battery plant owners to conduct the cleanup, estimated to cost $91
million. The responsible corporations also agreed to reimburse EPA $13.5 million for past cleanup
and future oversight costs.
In 1992, the cleanup of the plant's interior and the recycling of the contaminated books on
the property were completed. Starting in 1993, East Foundry Cove was dredged and the
contaminated sediments were hauled away and treated.

Fig. 13. Towers Where Foundry Cove
Sediment was Dewatered and Processed
for Shipment
Prior to excavation a dike was
constructed around the marsh to limit
transport of sediments into the Hudson.
West Foundry Cove was not dredged
since the contamination was less severe
than in eastern part of the cove and will
naturally be covered by sedimentation
over time. Constitution Marsh in
nearby South Cove was not excavated because it received only low levels of metals and since it is
a National Audubon Refuge human intervention was deemed an unnecessary risk.

Fig. 14. Aerial photograph of the site during
restoration. Note newly dug creeks to
maintain flushing and oxygenation. Also note
the ring around the site. This was a large
rubber bladder that resembled an inner tube.
It protected the site from incursions of water
as the cleanup and sediment removal
proceeded.
Remediation of metal contaminated sediments has important policy implications. Dredging
projects produce contaminated sediments which must be disposed and disposal of dredged
materials is often problematic. Often such materials are disposed of in sealed underground vaults,
but leaching of toxic materials from such vaults has often occurred, often with drastic
consequences, i.e. contamination of the groundwater supply. Since such a remedy was used in
Foundry Cove in 1971, groundwater contamination will be monitored for the next 30 years.
 Fig 15. Railcars used to transport sediment
from Foundry Cove (Cove is in foreground)
Fig 15. Railcars used to transport sediment
from Foundry Cove (Cove is in foreground)
A rail spur was also built to haul
away treated soil from East Foundry
Cove; removal by trucks would have
disrupted Cold Spring's historic
downtown district and potentially
damaged foundations to historic
buildings. East Foundry Cove cleanup
was completed in 1995.

Fig. 16. Foundry Cove Marsh, after
replanting, June 1995
The top layer of contaminated soil was removed from nearby residential yards and
re-landscaped. Wetland replanting efforts have just recently been completed, and the battery plant
and processing towers are coming down; the site will soon be an empty lot. Wetland recovery will
be monitored for a number of years.
The Hudson River serves as an example of how ecosystem contamination may have broad
implications for long-term ecological and economic sustainability. Since the large scale release of
Cd and other metals into the Hudson River more than 30 years ago, only recently has the issue of
how to clean up the Hudson been resolved. PCB's entered the Hudson about 20 years ago and the
issue of how to clean this contamination is still unresolved. Toxic contaminants have led to health
concerns, a reduced commercial fishery, debate over the appropriate course for cleaning the river,
and damage to the Hudson's public image.
Protecting and managing natural resources and promoting economic growth no longer
occupy separate interests. Policy-makers are now realizing the interrelatedness of environmental,
economic, and social programs, and balancing these concerns is no simple task. Understanding
how contaminants get into the environment, what happens to them once they are there, and what
can be done to minimize their impacts or remove them will be crucial to policy-makers forced to
balance known (and unknown) risks of contaminants with the need for development.
 Foundry Cove is situated in the Village of Cold Spring, in Putnam County, NY, approximately 54
miles north of Battery Park, NYC (Fig 1). Foundry Cove is a well defined inlet of the Hudson
River. This cove contains freshwater marshes and mud flats and is tidally influenced (Fig. 2). A
railroad trestle divides the cove into east and west; flow from the Hudson River into West
Foundry Cove is unrestricted, while flow into East Foundry Cove is restricted to a 65 foot wide
passage under the railroad
trestle. The water is
generally fresh but the
salinity may reach 2-6 parts
per thousand in periods of
low fresh water flow.
Foundry Cove is situated in the Village of Cold Spring, in Putnam County, NY, approximately 54
miles north of Battery Park, NYC (Fig 1). Foundry Cove is a well defined inlet of the Hudson
River. This cove contains freshwater marshes and mud flats and is tidally influenced (Fig. 2). A
railroad trestle divides the cove into east and west; flow from the Hudson River into West
Foundry Cove is unrestricted, while flow into East Foundry Cove is restricted to a 65 foot wide
passage under the railroad
trestle. The water is
generally fresh but the
salinity may reach 2-6 parts
per thousand in periods of
low fresh water flow.
 Fig. 2. Foundry Cove,
foreground, with Constitution
Marsh Audubon Sanctuary at
Center Left. South Cove is at the
Rear
Fig. 2. Foundry Cove,
foreground, with Constitution
Marsh Audubon Sanctuary at
Center Left. South Cove is at the
Rear
 sediments in East Foundry Cove in 1983
sediments in East Foundry Cove in 1983










 Fig 15. Railcars used to transport sediment
from Foundry Cove (Cove is in foreground)
Fig 15. Railcars used to transport sediment
from Foundry Cove (Cove is in foreground)